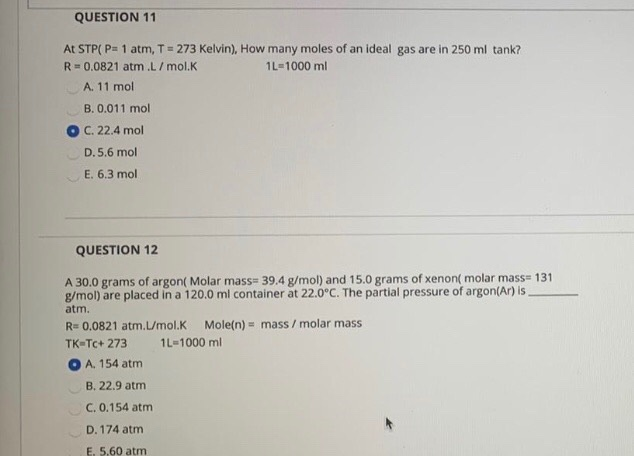
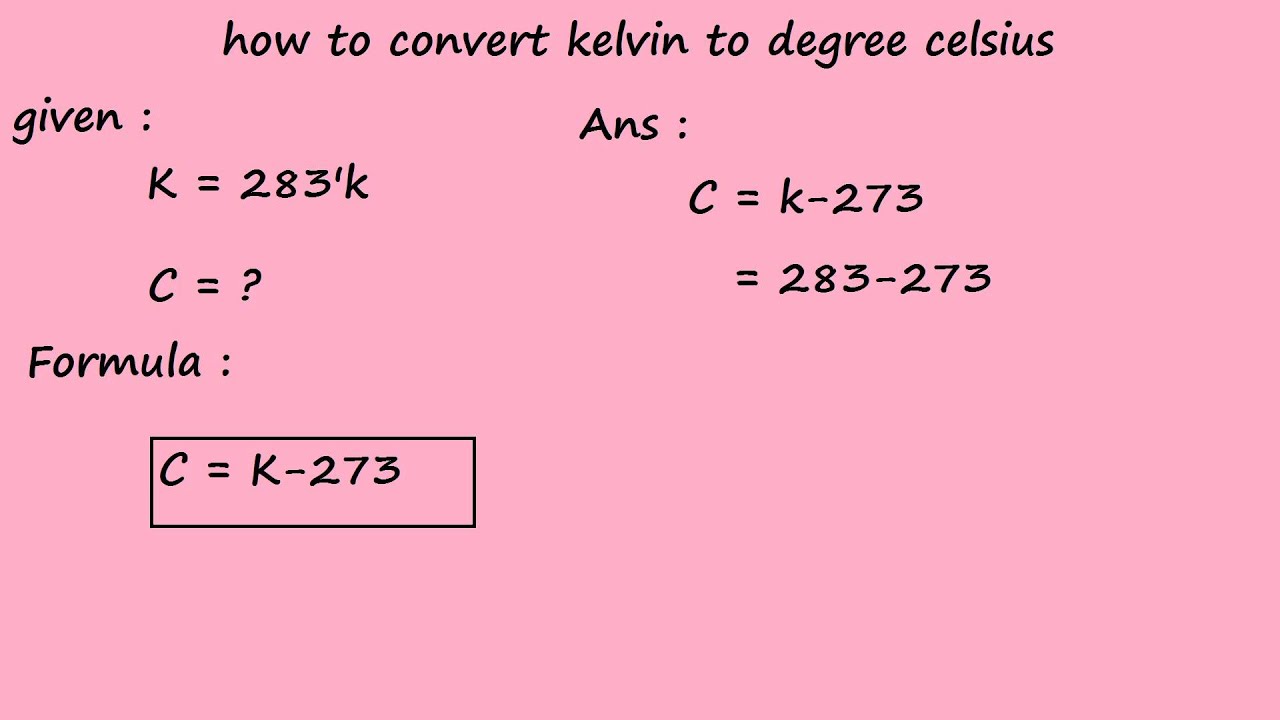The enthalpy of a reaction at 273 K. is -3.57 KJ. What will be the enthalpy of reaction at 373 K if DeltaC(p) = zero :-

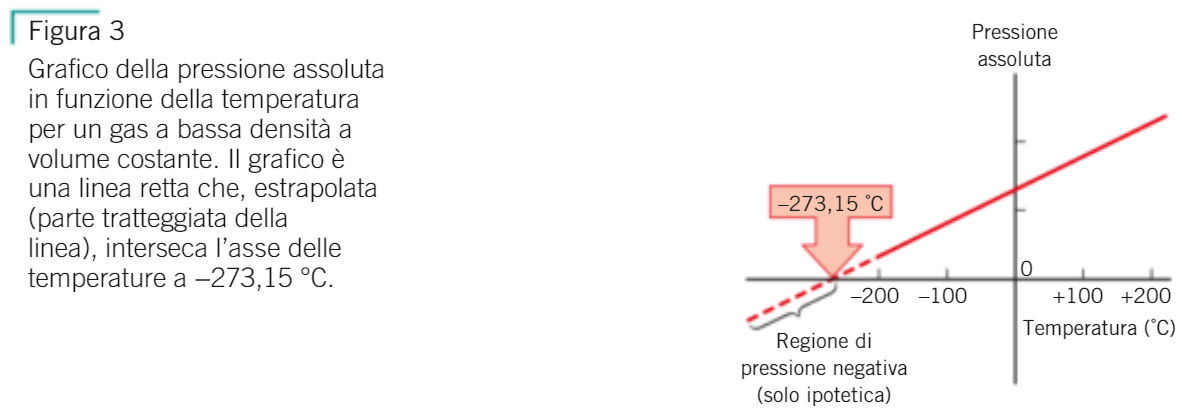
SVG, Vettoriale - Celsius, Kelvin, Fahrenheit Temperatura Conversione Scale Zero Assoluto è Della Scala Kelvin, -273 Sulla Scala Celsius E -459 Sulla Scala Fahrenheit. Image 15013057.
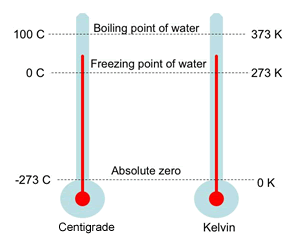
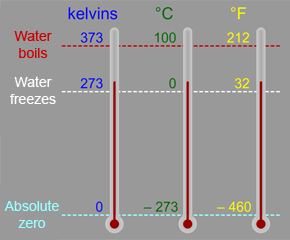
a What is the relationship between the Celcius and the Kelvin scales of temperature? b Convert i 273∘C ii 293 K to ∘C