![Optical absorption spectra of PCS fiber with incorporated [ n L-Co]Br2... | Download Scientific Diagram Optical absorption spectra of PCS fiber with incorporated [ n L-Co]Br2... | Download Scientific Diagram](https://www.researchgate.net/publication/299459070/figure/fig3/AS:1086757638537292@1636114620475/Optical-absorption-spectra-of-PCS-fiber-with-incorporated-n-L-CoBr2-reagent-The.jpg)
Optical absorption spectra of PCS fiber with incorporated [ n L-Co]Br2... | Download Scientific Diagram

SOLVED: The equilibrium constant; Kc for the decomposition of COBr2 COBrz(g) 2 co(g) Br2(g) is 0.190. What is Kc for the following reaction? 2COBrz(g) 2CO(g) 2Br2(g)
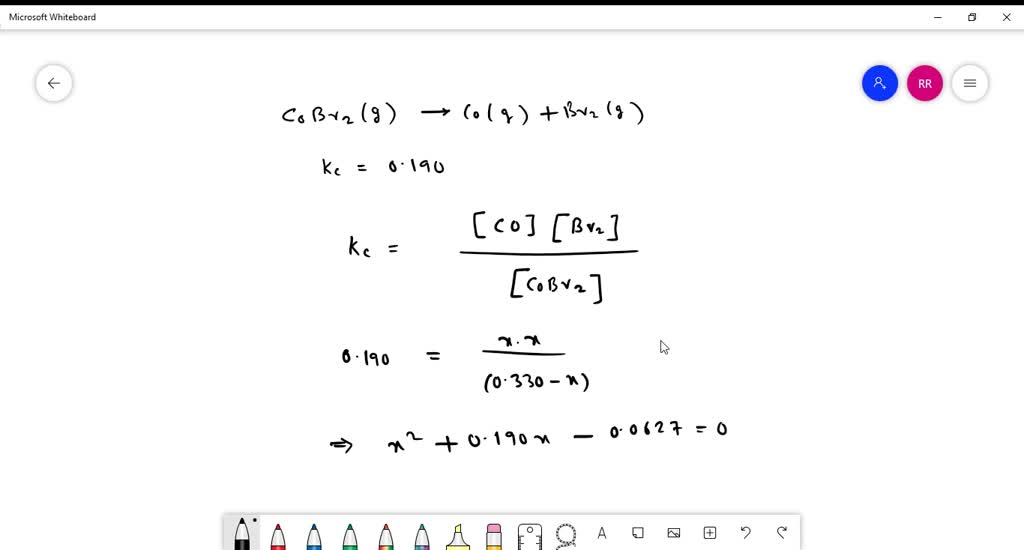
SOLVED: Carbonyl bromide decomposes to carbon monoxide and bromine. COBr2(g) –>. CO(g) + Br2(g) Kc is 0.190 at 73 °C. If an initial concentration of 0.330 M COBr2 is allowed to equilibrate,

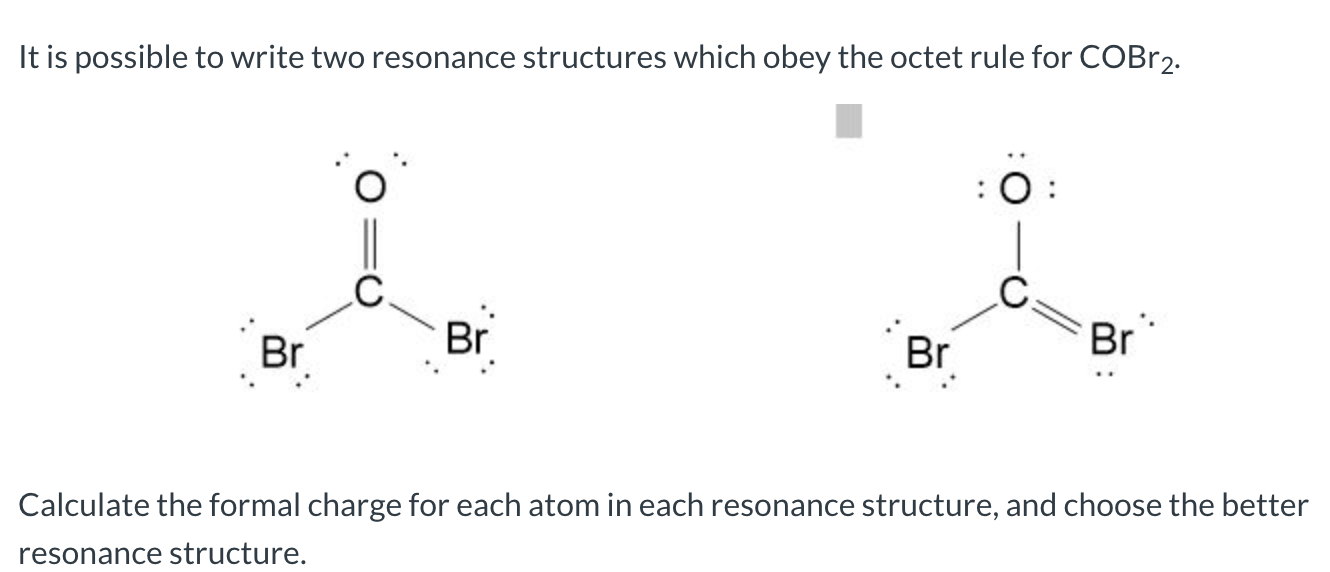
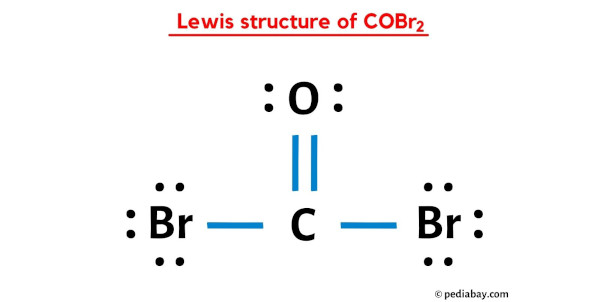
What is the Lewis Structure of COBr_2? (please explain steps and # of valence electrons used to find solution) | Homework.Study.com

Carbonyl bromide decomposes to carbon monoxide and bromine. COBr2(g) arrow CO(g) + Br2(g); Kc = 0.19 at 73 degrees Celsius If an initial concentration of 0.63 M COBr2 is allowed to equilibrate,





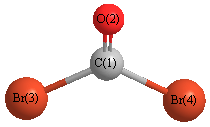
![Solved] The product obtained in the reaction of Mn2(CO)10 with Br2 i Solved] The product obtained in the reaction of Mn2(CO)10 with Br2 i](https://storage.googleapis.com/tb-img/production/22/01/F1_Utakarsha_13-01-22_Savita_D20.png)



![OneClass: What are the isomers of [CoBr2(en)(NH3)2][BF4]? OneClass: What are the isomers of [CoBr2(en)(NH3)2][BF4]?](https://prealliance-textbook-qa.oneclass.com/qa_images/homework_help/question/qa_images/121/12105729.webp)

