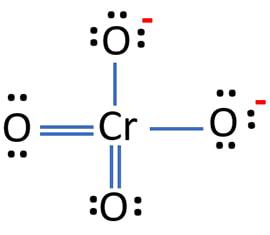
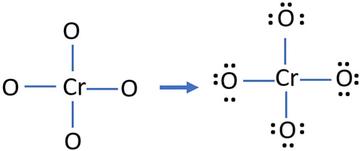
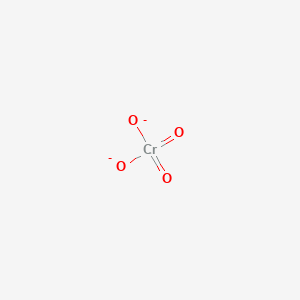
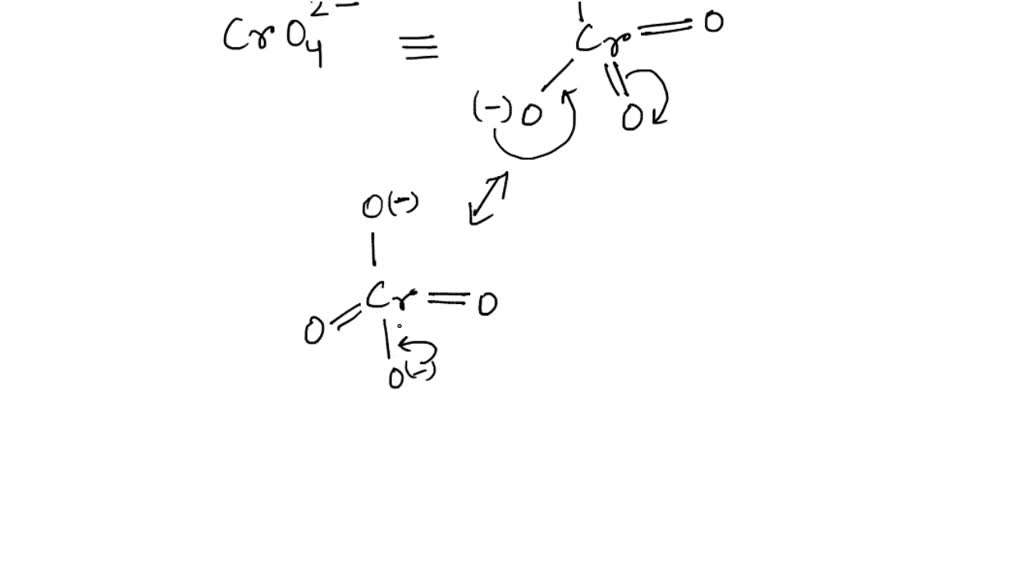
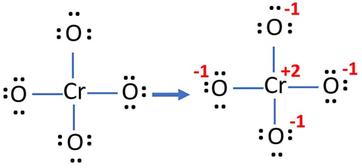
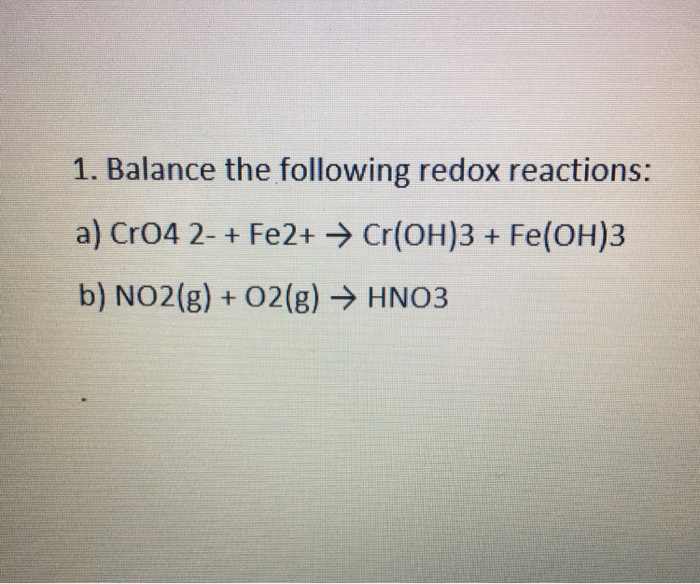Oxidation Number for CrO4 2- . Oxidation state of Chromate ion. Oxidation state of cro4 2-. Cro42- - YouTube
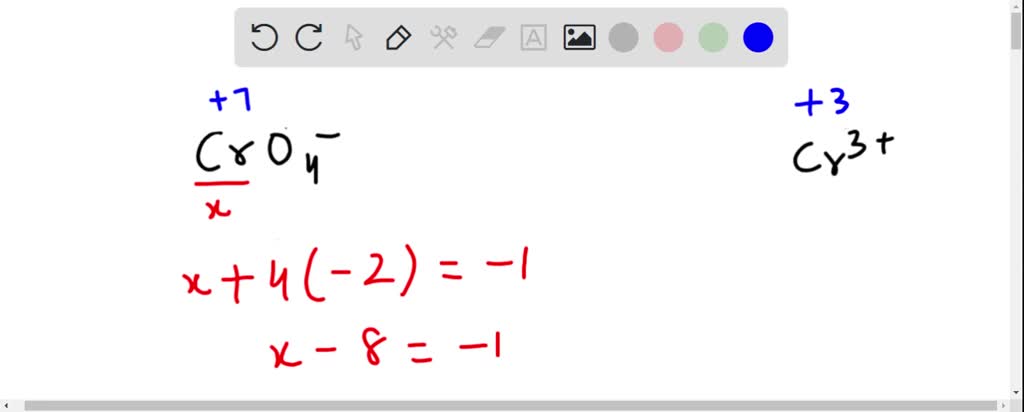
SOLVED: How many electrons does the chromium atom in the chromate ion, CrO4-(aq) , gain when it is converted to the Cr3+(aq) ion? a) 2 b) 3 c) 4 d) 5 e)

SO2(g) + ....O2(g)→ ...SO3(g) According to the above unbalanced equation, how many moles of SO2(g) will react completely with 1 mole of O2(g) ?

![Balance Redox Rxns: Fe(OH)3 + [Cr(OH)4]-1 Fe(OH)2 + CrO ppt download Balance Redox Rxns: Fe(OH)3 + [Cr(OH)4]-1 Fe(OH)2 + CrO ppt download](https://slideplayer.com/16529329/96/images/slide_1.jpg)