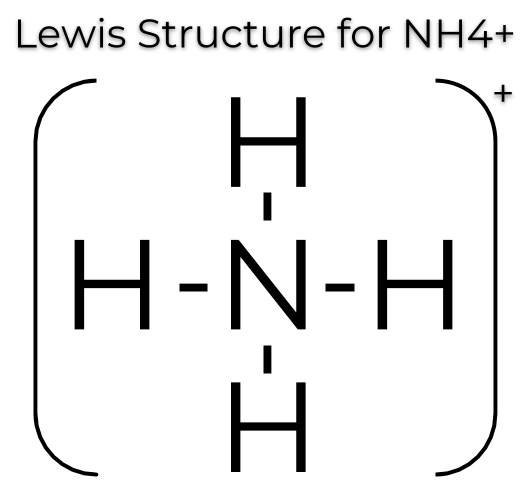
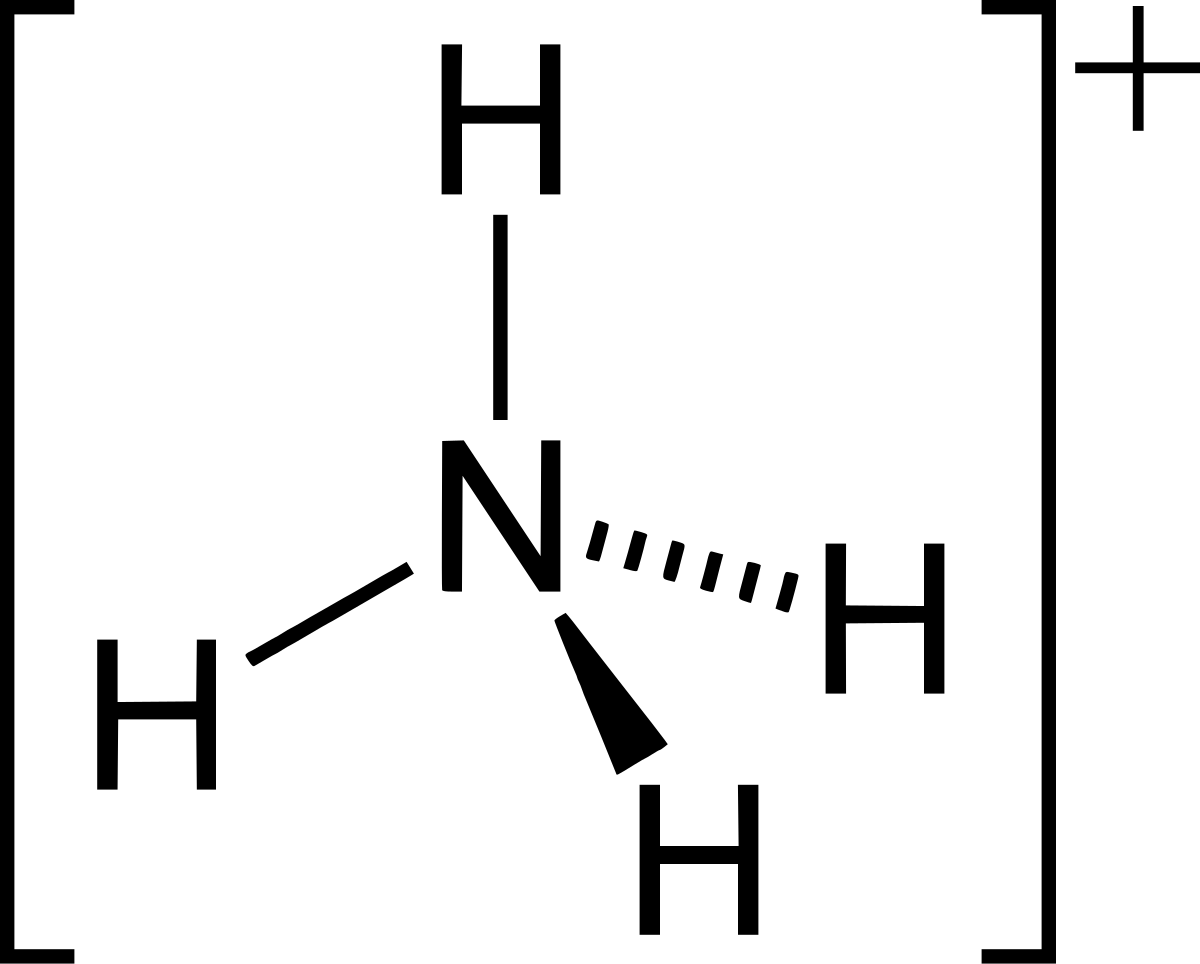
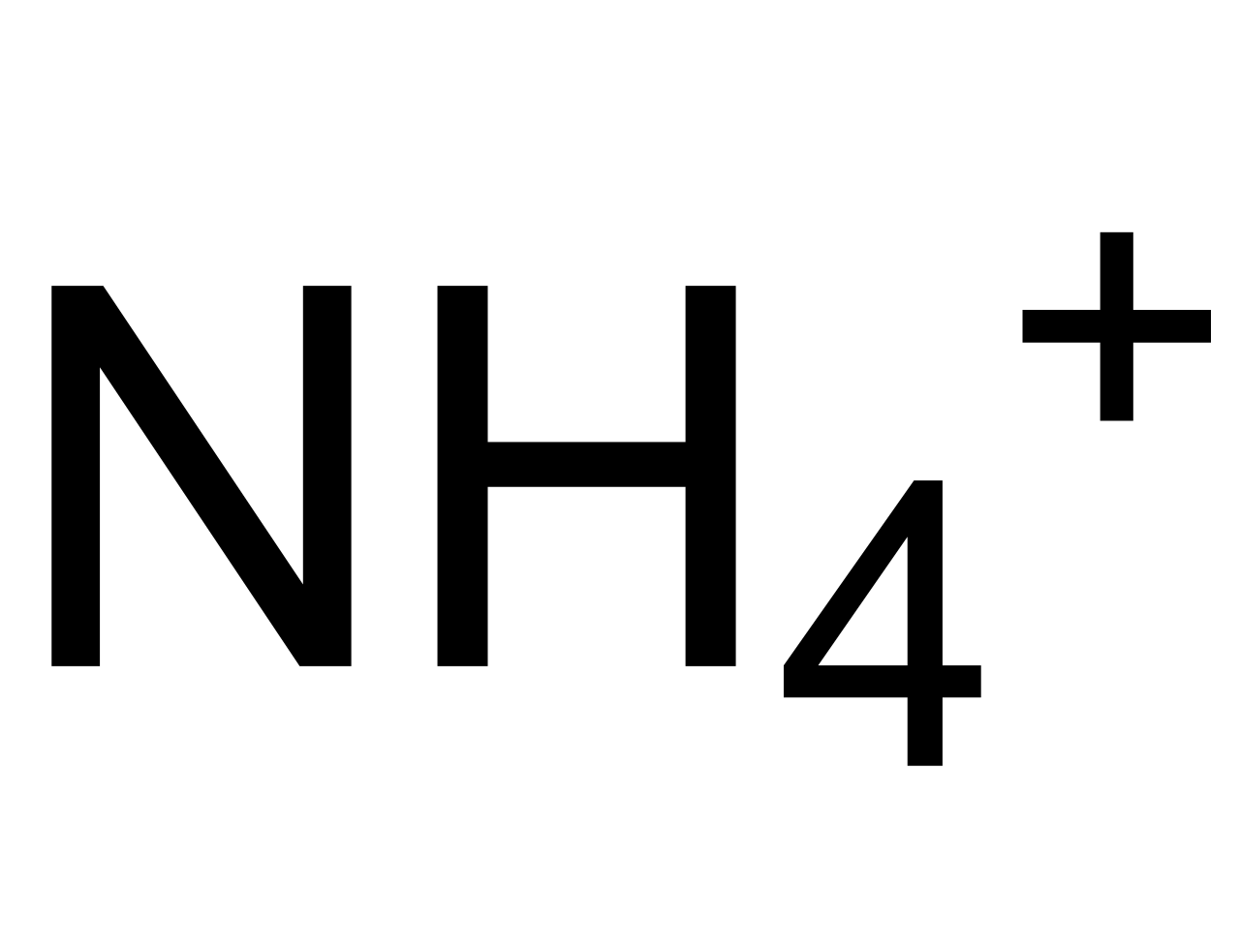
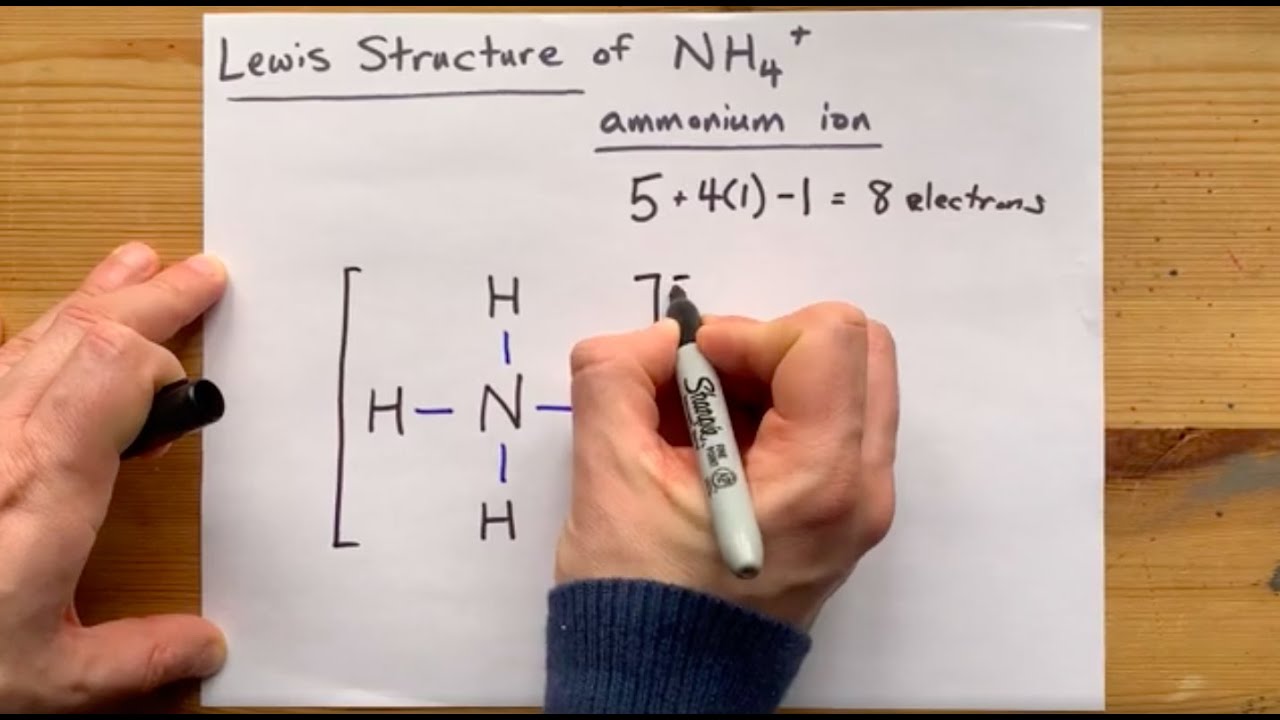
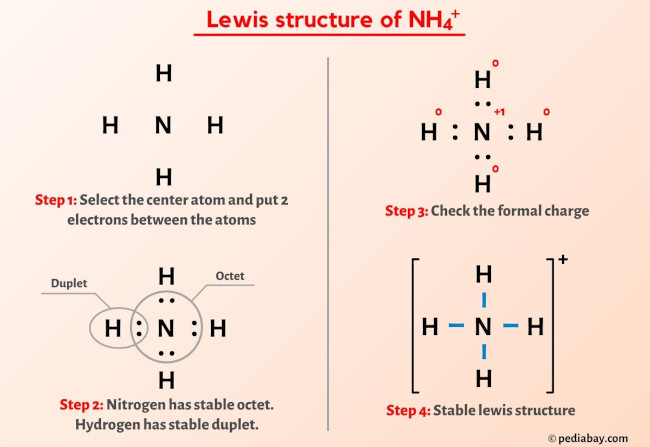
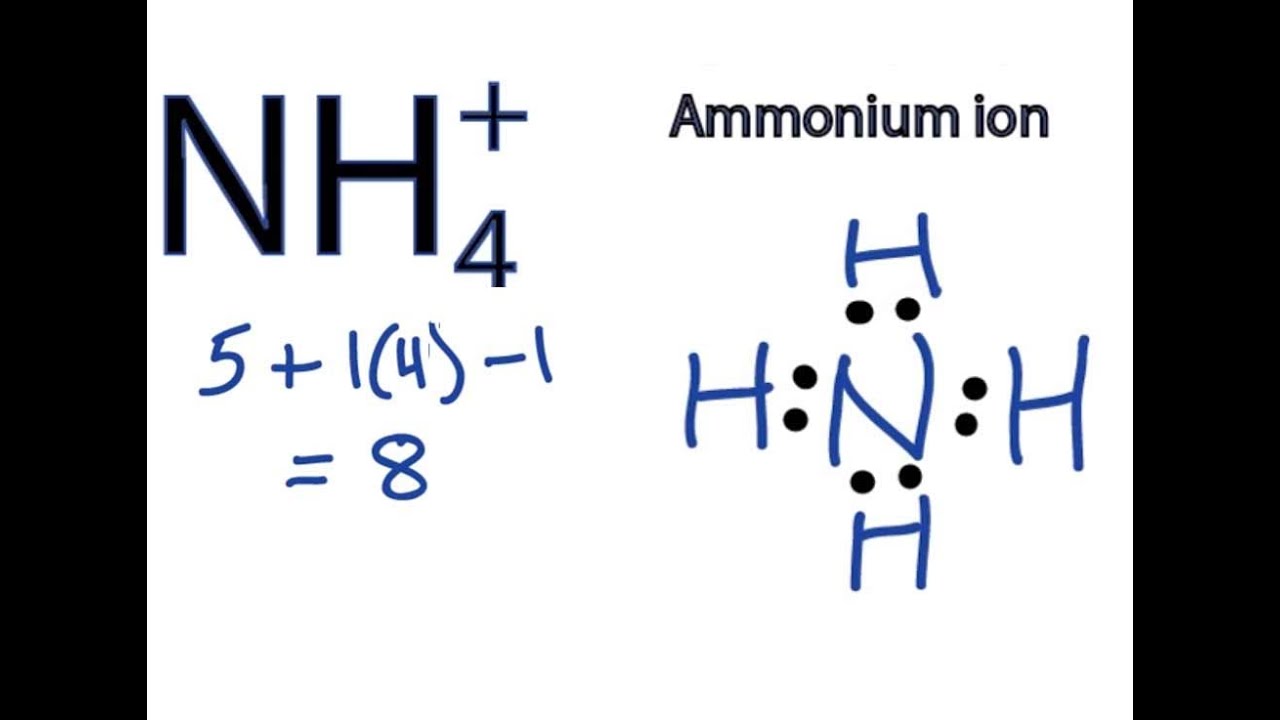
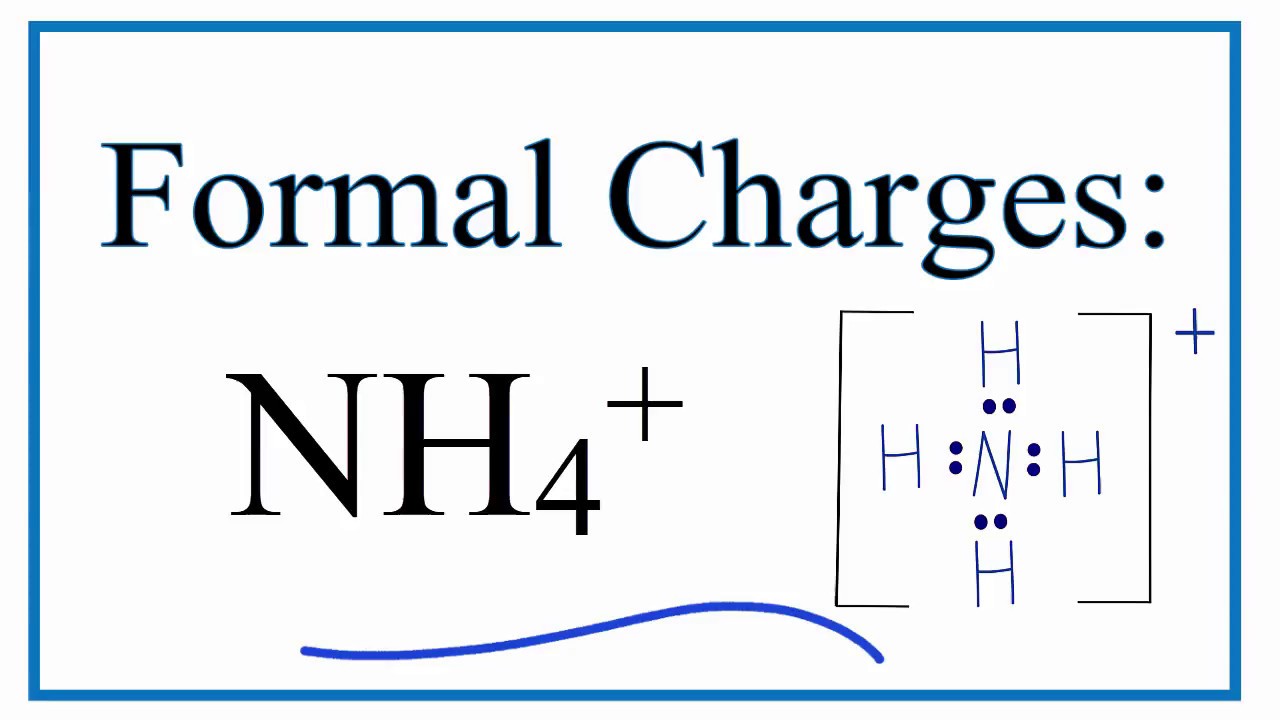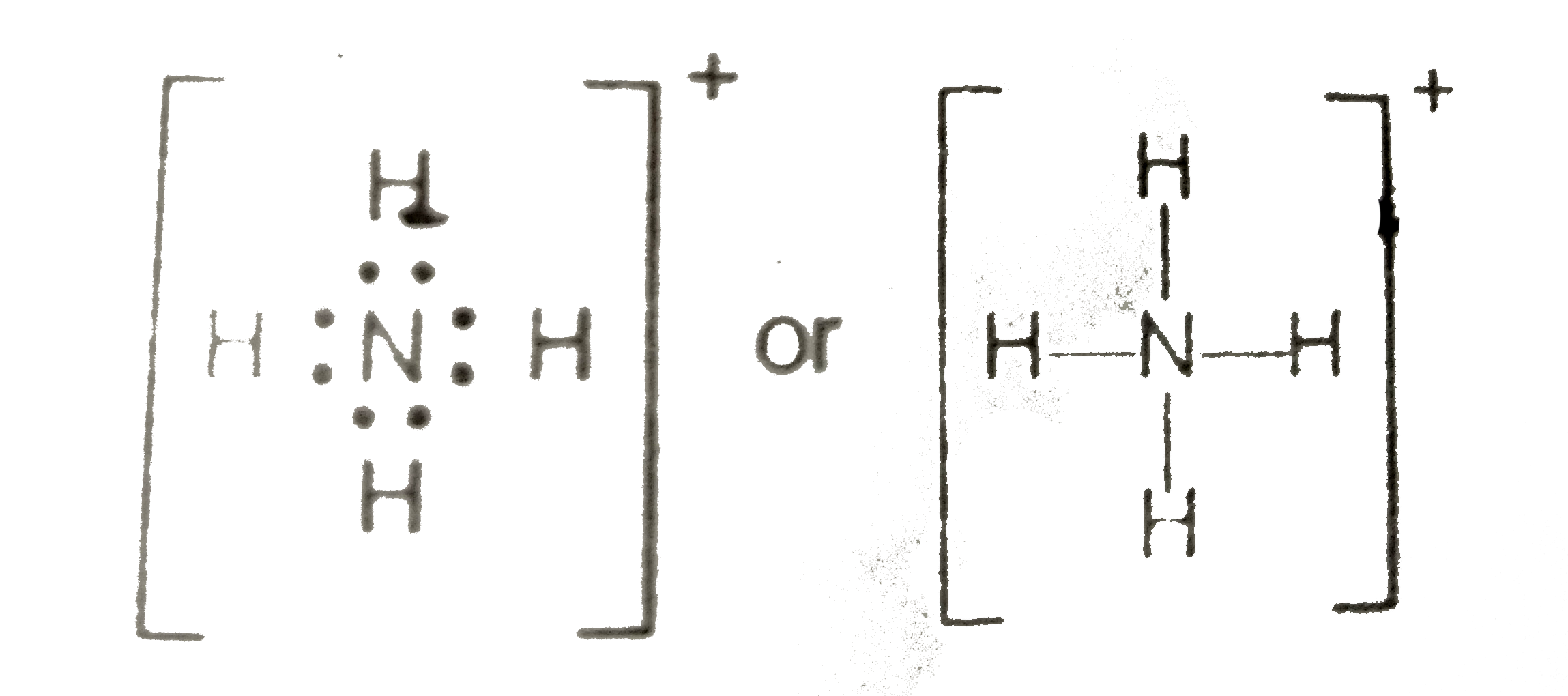Ammonium ion NH4+ - Explanation, Properties of Ammonium ion, Preparation and some Applications along with FAQs
![Draw an electron dot diagram to show the formation of ammonium ion [Atomic number: N = 7 and H = 1 ]. Draw an electron dot diagram to show the formation of ammonium ion [Atomic number: N = 7 and H = 1 ].](https://haygot.s3.amazonaws.com/questions/612389_578209_ans.png)
Draw an electron dot diagram to show the formation of ammonium ion [Atomic number: N = 7 and H = 1 ].

Immagini Stock - Il Catione Ammonio è Uno Ione Poliatomico Caricato Positivamente Con La Formula Chimica Nh4 +. Esso è Formato Dalla Protonazione Di Ammoniaca (nh3). Image 26350795.
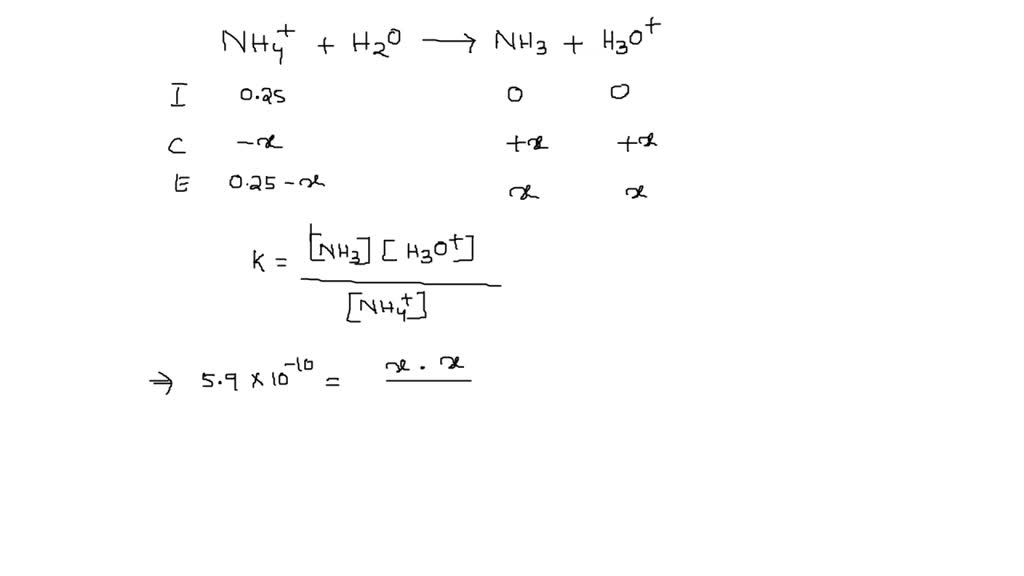
SOLVED: The value of the ionization constant for ammonium ion (NH4+) is 5.9 × 10−10. What is the pH of 0.25 M NH4+(aq)? a. 4.92 b. 9.83 c. 5.22 d. 9.23 e. 0.60

Ammonium ion NH4+ - Explanation, Properties of Ammonium ion, Preparation and some Applications along with FAQs