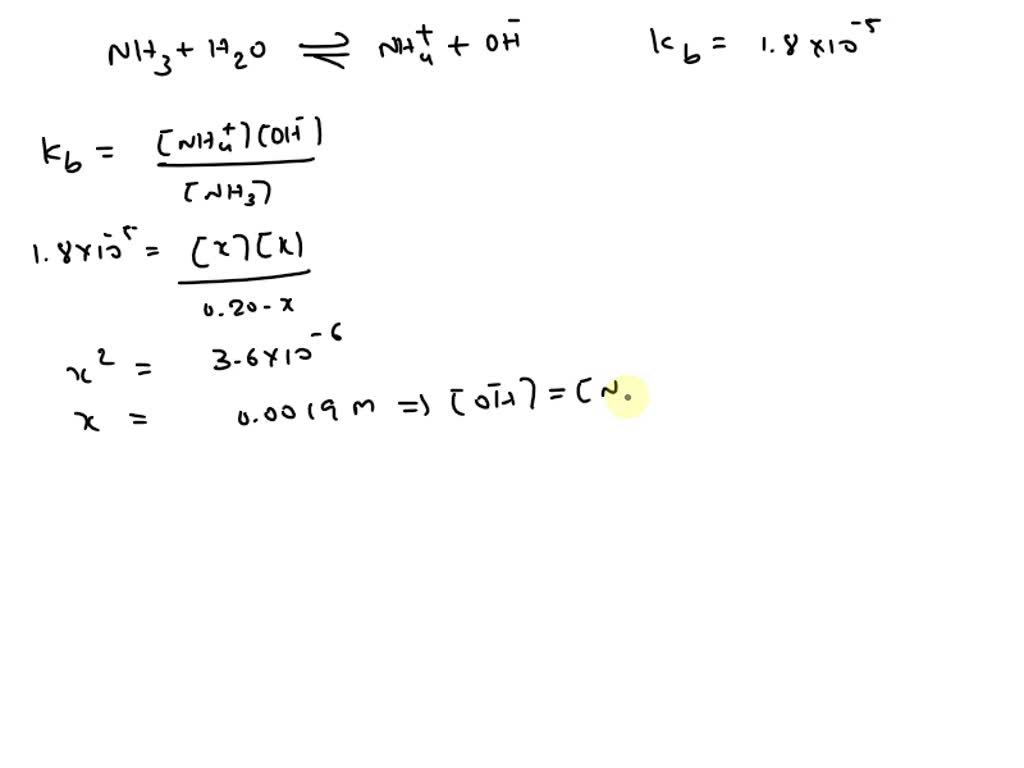Calculate [OH^– ] in 0.20 M solution of NH3 if Kb for NH3 is 1.8 × 10^–5 . - Sarthaks eConnect | Largest Online Education Community

NH3 is a weak base (Kb = 1.8 times 10^-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.050 M in
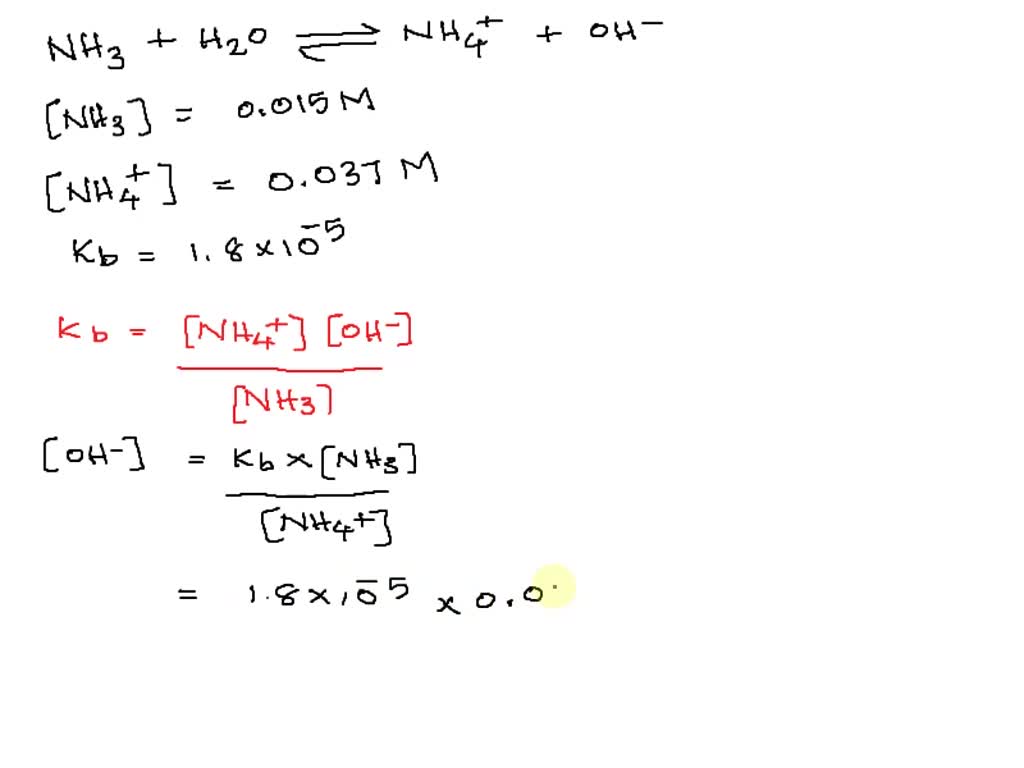
SOLVED: Calculate the pH of a buffer solution consisting of 0.051 M NH3 and 0.037 M NH4+. The Kb for NH3 = 1.8 x 10-5.

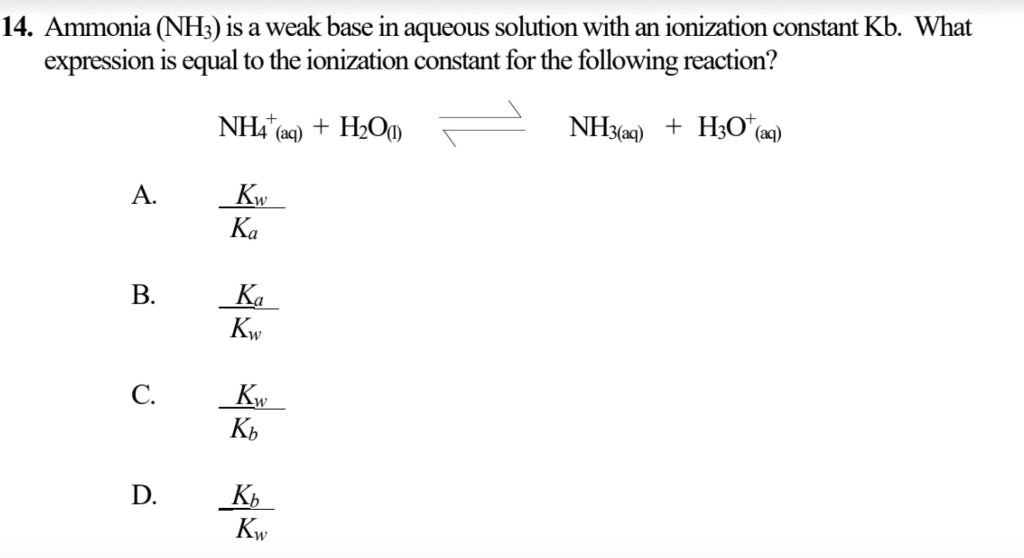
Kb for NH3=1.8xx10^(-5) at 25^(@)C. What is the value of Ka for NH4^(+) ion at the same temperature ?
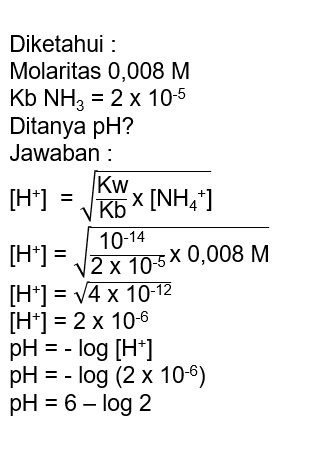
the concentration of NH_4+ IONS in an aqueous solution which is 0.02M NH3 0.01M KOH would be: kb=1.8×10^ 5

L'effetto ione comune Un acido debole o una base debole ionizzeranno parzialmente in soluzione acquosa. Per esempio, Gli acidi deboli hanno usualmente. - ppt scaricare

NH3 is a weak base (Kb = 1.8 times 10^-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.050 M in

SciELO - Brasil - HYDROPHOBIC MEMBRANE TECHNOLOGY FOR AMMONIA EXTRACTION FROM WASTEWATERS HYDROPHOBIC MEMBRANE TECHNOLOGY FOR AMMONIA EXTRACTION FROM WASTEWATERS