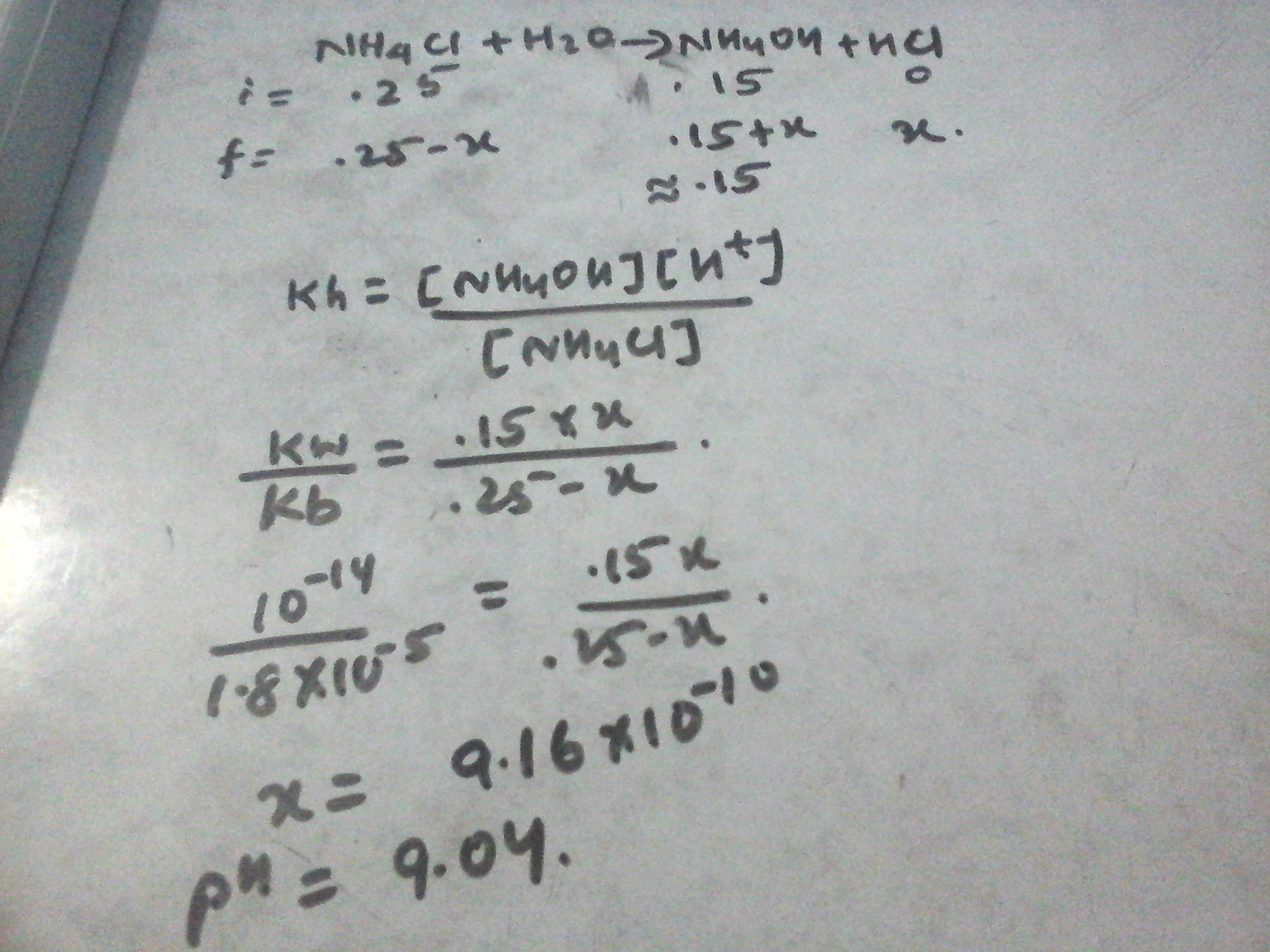Welcome to Chem Zipper.com......: What will be the pH of the buffer solution containing 0.15 moles of NH4OH and 0.25 moles of NH4Cl Kb for NH4OH is 1.8x10^-5.
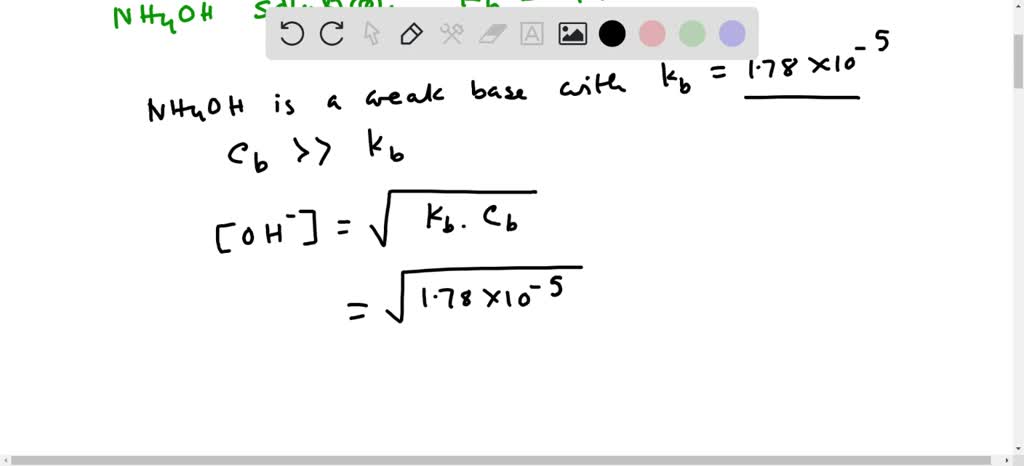
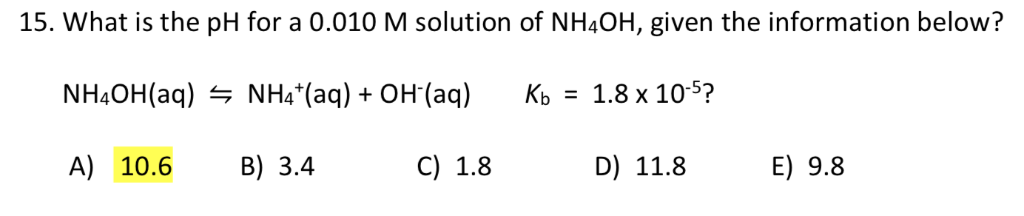
SOLVED: Answer the following equation:Ammonium Hydroxide, NH4OH is a weak base. Calculate the pH of 0.60 M solution of ammonium hydroxide. Kb= 1.78 x 10^-5

SOLVED: 1. What is the percent ionization of NH4OH in the in a 0.18 M solution of NH4OH? (Kb = 1.8 x10-5) 2. What is the concentration of hydroxide ions (OH-) in
Welcome to Chem Zipper.com......: Calculate the emf of the cell: Pt H2(1atm)ICH3COOH(0.1M) II NH4OH(0.01M)IH2(1 atm)Pt and Ka for CH3COOH= 1.8x10^-5 and Kb for NH4OH = 1.8x10^-5