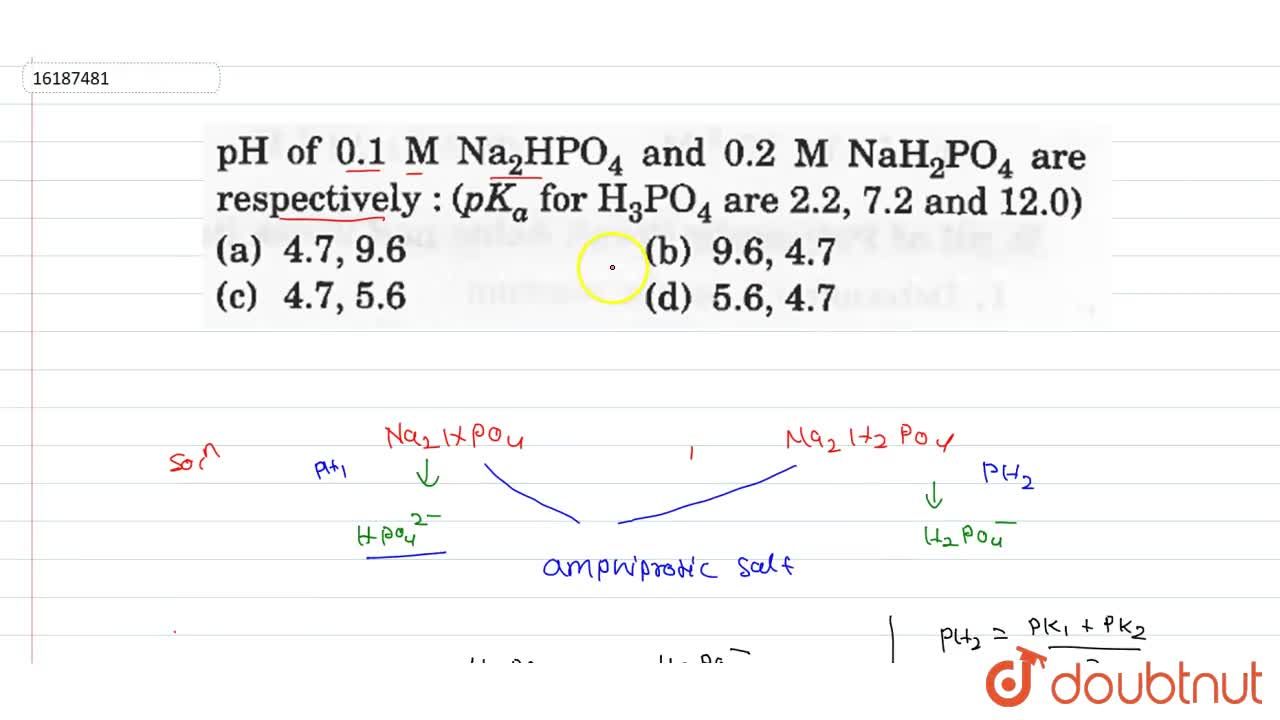
SOLVED: What is the pH of a buffer that consists of 0.20 M NaH2PO4 and 0.40 M Na2HPO4? For H2PO4–, Ka = 6.2 × 10–8. Select one: a. 6.51 b. 7.51 c. 8.13 d. 7.90 e. 6.91
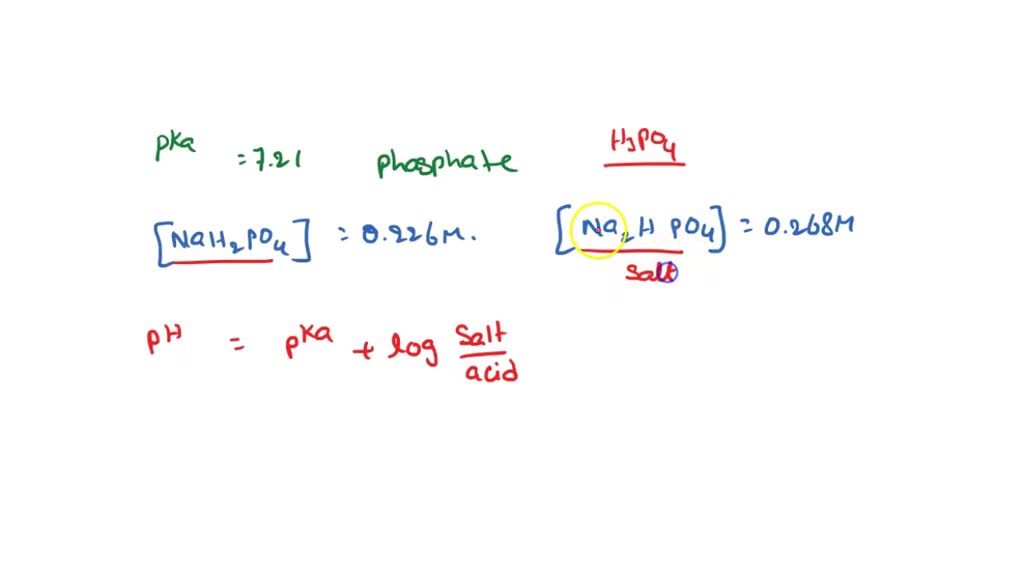
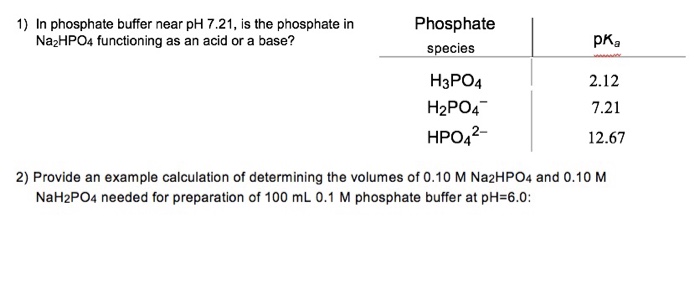
SOLVED: 1. Using the pKa of (7.21) phosphate (phosphate has 3 pKas, which one is relevant) and the concentrations of NaH2PO4 (.226) and Na2HPO4 (.268). Calculate the pH of the buffer.

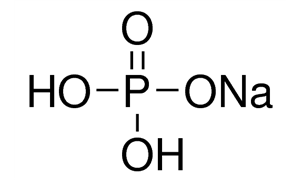
TRACCIA 9 Calcolare il pH di una soluzione ottenuta sciogliendo 12 g di NaH2PO4 in 100 ml di KOH 2 M. (H3PO4: Ka1=7,6x10-3, Ka2=6,2x10-8, Ka1=4,4x10-13). - ppt scaricare

If 2.5 moles each of H3PO4,NaH2PO4,Na2HPO4 and Na3PO4 are mixed together to form an aqueous solution, then the resulting pH is:Given values of Ka are: Ka1 = 10^-3 Ka2 = 10^-7 Ka3 = 10^-13
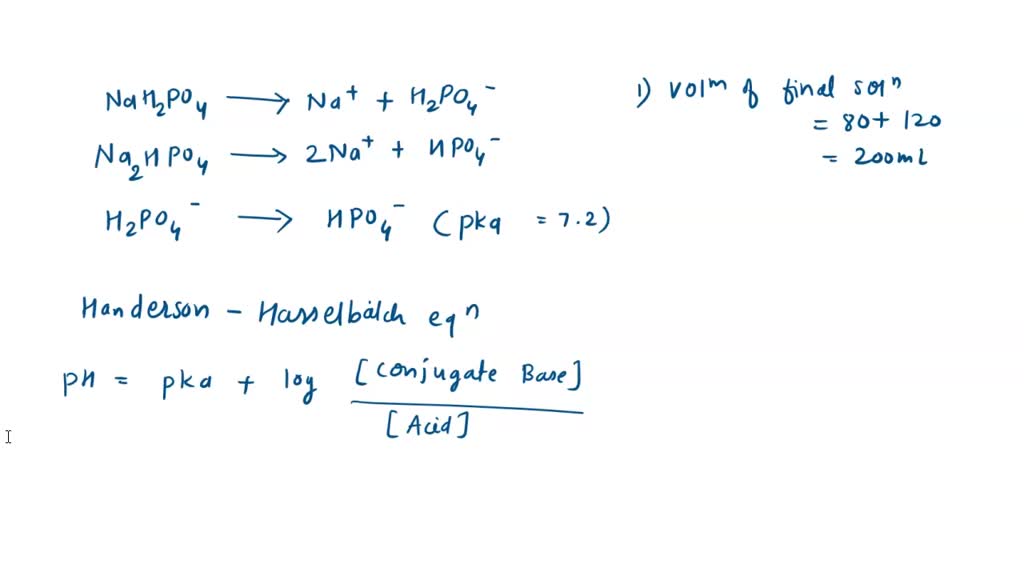
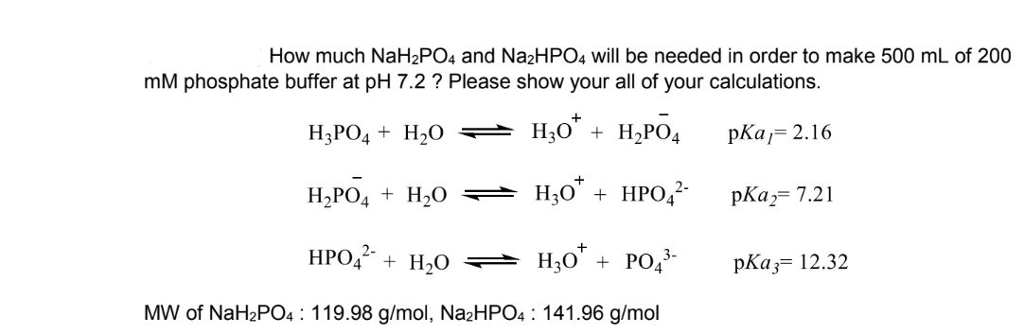
SOLVED: Calculate the pH of a buffer solution that is made by mixing 80 mL of 0.6 M NaH2PO4 with 120 mL of 0.8 M of Na2HPO4. Calculate the pH when 10


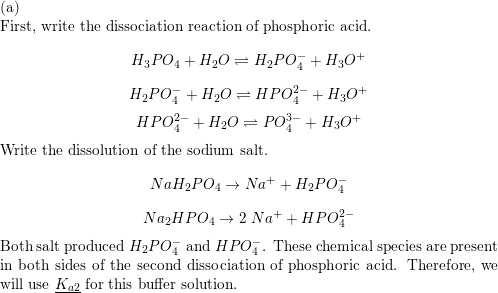



![Answered: [A-] pH= pka + log [HA] a. Determine… | bartleby Answered: [A-] pH= pka + log [HA] a. Determine… | bartleby](https://content.bartleby.com/qna-images/question/0efbec1c-3aeb-47c8-90fa-39b6de84bb74/c728bc90-2d81-4d3c-b78c-7dd9d14854f6/k04zzb.jpeg)